Experts Who Make the Impossible Possible
Think Bold. Think Scientific.
Think Clindus.
with excellence and precision.

Research
About Clindus
Clindus Research Solutions is a leading clinical research partner committed to advancing healthcare innovation. Established in 2023 and headquartered in Bengaluru, Karnataka, India, we specialize in tailored clinical research services for pharmaceutical, biotech, and healthcare organizations across the globe. Our focus is on delivering high-quality, compliant, and cost-effective research solutions that elevate client success and accelerate new product development in the healthcare ecosystem.
- Certified Expertise
Clindus is a leading SMO, expertly navigating regulatory complexities while ensuring full compliance with ICH-GCP and NDCT guidelines.
- Expert Experience
Our experienced investigators and coordinators deliver precise, timely clinical trials, ensuring high-quality data and effective patient recruitment across therapeutic areas.
- Comprehensive Services
From Phase I–IV trials to BA/BE studies and post-marketing surveillance, we deliver comprehensive clinical research support across disciplines.
- Academy & Training
As your extended team, we manage trials and develop talent through Clindus Academy, delivering practical training and future-ready certifications.
Research
About Clindus
Clindus Research Solutions is a leading clinical research partner committed to advancing healthcare innovation. Established in 2023 and headquartered in Bengaluru, Karnataka, India, we specialize in tailored clinical research services for pharmaceutical, biotech, and healthcare organizations across the globe. Our focus is on delivering high-quality, compliant, and cost-effective research solutions that elevate client success and accelerate new product development in the healthcare ecosystem.
- Certified Expertise
Clindus is a leading SMO, expertly navigating regulatory complexities while ensuring full compliance with ICH-GCP and NDCT guidelines.
- Expert Experience
Our experienced investigators and coordinators deliver precise, timely clinical trials, ensuring high-quality data and effective patient recruitment across therapeutic areas.
- Comprehensive Services
From Phase I–IV trials to BA/BE studies and post-marketing surveillance, we deliver comprehensive clinical research support across disciplines.
- Academy & Training
As your extended team, we manage trials and develop talent through Clindus Academy, delivering practical training and future-ready certifications.
Research
About Clindus
Clindus Research Solutions is a leading clinical research partner committed to advancing healthcare innovation. Established in 2023 and headquartered in Bengaluru, Karnataka, India, we specialize in tailored clinical research services for pharmaceutical, biotech, and healthcare organizations across the globe. Our focus is on delivering high-quality, compliant, and cost-effective research solutions that elevate client success and accelerate new product development in the healthcare ecosystem.
- Certified Expertise
Clindus is a leading SMO, expertly navigating regulatory complexities while ensuring full compliance with ICH-GCP and NDCT guidelines.
- Expert Experience
Our experienced investigators and coordinators deliver precise, timely clinical trials, ensuring high-quality data and effective patient recruitment across therapeutic areas.
- Comprehensive Services
From Phase I–IV trials to BA/BE studies and post-marketing surveillance, we deliver comprehensive clinical research support across disciplines.
- Academy & Training
As your extended team, we manage trials and develop talent through Clindus Academy, delivering practical training and future-ready certifications.

Our Services
Strategic Research Solutions
We offer professional services tailored to support clinical research and healthcare organizations:
Site Management
Services
Let us be your clinical trial superhero!
Our site management services take care of the details, so you can focus on what really matters.
Medical
Writing
We turn medical jargon into engaging content that speaks to your audience, whether it’s patients, physicians, or the regulatory agencies.
Medical
Monitoring
Expert-led medical monitoring delivering real-time safety surveillance, protocol adherence, and regulatory-compliant risk assessment.
Clindus
Academy
As an ‘Extended Arm of Your Team,’ we don’t just manage trials; we build talent ensuring that the clinical research workforce is always future-ready.
Therapeutic
Focus Services

Dermatology

Gynecology

Oncology

Urology

Opthalmology

Gastroenterology

Cardiovascular Disease

Infectious Disease

Pain and Surgery

Rare Disease

Neuropsychiatry
Respiratory Medicine
Therapeutic
Focus Services

Dermatology

Gynecology

Oncology

Urology

Opthalmology

Gastroenterology

Cardiovascular Disease

Infectious Disease

Pain and Surgery

Rare Disease

Neuropsychiatry

Respiratory Medicine
Reach
Our Operation
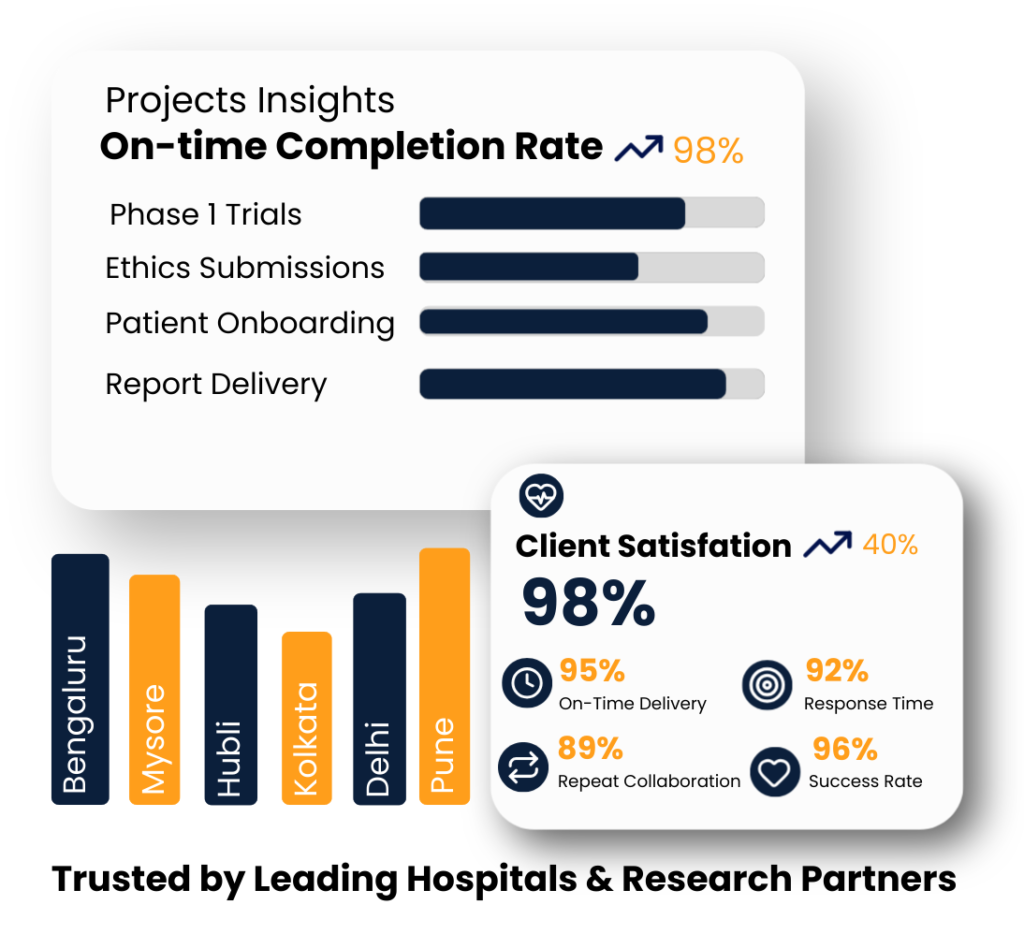
Our services extend across eight vibrant cities, weaving a network of care and innovation to empower communities everywhere.

Mysore

Bengaluru

Delhi

MP

Kolkata

Pune

Hubli -Dharwad

Jaipur
Research
Why Choose Us
At Clindus Research Solutions, we deliver excellence across all phases of clinical trial management, combining precision, innovation, and tailored approaches. Our expert team leverages advanced technology to ensure efficient operations, reduce risks, and accelerate patient recruitment, consistently driving high-quality outcomes and reliable results for every study.
01. End-to-End Operational Excellence
We handle clinical trials from start to finish, including feasibility analysis, protocol development, patient recruitment, and trial management. Our holistic approach ensures that no detail is overlooked from site selection to final closeout.
02. Adaptive & Scalable Methodologies
Whether it is a small-scale trial or a multinational study, we provide flexible, scalable solutions tailored to meet your specific needs. We adapt our resources and strategies to align with the unique demands of your protocol.
03. Proactive Risk & Compliance Oversight
We focus on minimizing risks with robust contingency plans, ensuring smooth operations even in challenging situations. Our proactive monitoring identifies potential bottlenecks before they impact your study timelines.
01. End-to-End Operational Excellence
We handle clinical trials from start to finish, including feasibility analysis, protocol development, patient recruitment, and trial management. Our holistic approach ensures that no detail is overlooked from site selection to final closeout.
02. Adaptive & Scalable Methodologies
Whether it is a small-scale trial or a multinational study, we provide flexible, scalable solutions tailored to meet your specific needs. We adapt our resources and strategies to align with the unique demands of your protocol.
03. Proactive Risk & Compliance Oversight
We focus on minimizing risks with robust contingency plans, ensuring smooth operations even in challenging situations. Our proactive monitoring identifies potential bottlenecks before they impact your study timelines.

Testimonials
Results Our Clients Love
Entering private practice, I never considered research participation. Clindus made it seamless by managing studies, handling regulatory processes, and providing support staff, enabling access to advanced therapies for my patients.
Clindus ensured comprehensive site efficiency through robust monitoring frameworks and structured processes. Their expertise effectively minimized operational bottlenecks, improving workflow, enhancing study timelines, and maintaining high standards of quality and compliance.
Recognition
We are proudly recognized by certifications and bodies




Unleash Your Business Potential with
CLINDUS
Partner with us today for unparalleled service and success.
Clindus Research Solutions is a trusted clinical research partner dedicated to driving innovation in healthcare. Founded in 2023 and headquartered in Bengaluru, Karnataka, India, we provide customized clinical research services to pharmaceutical, biotechnology, and healthcare organizations worldwide.
